|
I can see that once the renovation has been done, as we look at doing all the AP's and stuff it would be a good idea to do a WiFi survey. The main office is also currently going through a major renovation so in about 2 months the whole layout will be dramatically different. Lactation: Because of the potential risk for serious adverse reactions in breastfed children, advise women not to breastfeed during treatment with LUTATHERA and for 2.5 months after the last dose.Just started my first network admin job and need advise! One thing I immediately notice here is that people often complain about WiFi coverage/speed/performance etc and it's difficult to pin down whats what. Avoid repeated administration of high doses of glucocorticoids during treatment with LUTATHERA. Glucocorticoids can induce downregulation of subtype 2 somatostatin receptors. Administer short- and long-acting octreotide during LUTATHERA treatment as recommended. Discontinue long-acting somatostatin analogs at least 4 weeks and short-acting octreotide at least 24 hours prior to each LUTATHERA dose. Somatostatin and its analogs competitively bind to somatostatin receptors and may interfere with the efficacy of LUTATHERA. Patients should be counseled and monitored in accordance with the LUTATHERA Prescribing Information. Renal Toxicity: In ERASMUS, 8 patients (4 years after treatment with LUTATHERA: myelodysplastic syndrome (2%), acute leukemia (1%), renal failure (2%), hypotension (1%), cardiac failure (2%), myocardial infarction (1%), and neuroendocrine hormonal crisis (1%).The median time to onset was 29 months (range, 9-45 months) for MDS and 55 months (range, 32-125 months) for acute leukemia. In ERASMUS, a phase 2 clinical study, 16 patients (2.0%) developed MDS and 4 (0.5%) developed acute leukemia. Secondary Myelodysplastic Syndrome and Leukemia: In NETTER-1, with a median follow-up time of 76 months in the main study, myelodysplastic syndrome (MDS) was reported in 2.3% of patients receiving LUTATHERA with long-acting octreotide compared with no patients receiving high-dose long-acting octreotide.Withhold dose, reduce dose, or permanently discontinue LUTATHERA based on the severity of myelosuppression. Among these 15 patients, 5 improved to grade 1, 9 to grade 2, and 1 to grade 3. Fifteen of the 19 patients in whom platelet recovery was not documented had post-nadir platelet counts. The median time to platelet recovery was 2 months. Of the 59 patients who developed thrombocytopenia, 68% had platelet recovery to baseline or normal levels. In NETTER-1, platelet nadir occurred at a median of 5.1 months following the first dose. Myelosuppression: In the NETTER-1 clinical trial, myelosuppression occurred more frequently in patients receiving LUTATHERA with long-acting octreotide compared with patients receiving high-dose long-acting octreotide (all grades/grade 3/4): anemia (81%/0 vs 54%/1%), thrombocytopenia (53%/1% vs 17%/0), and neutropenia (26%/3% vs 11%/0).Minimize radiation exposure to patients, medical personnel, and household contacts during and after treatment with LUTATHERA consistent with institutional good radiation safety practices, patient management procedures, Nuclear Regulatory Commission patient release guidance, and instructions to the patient for follow-up radiation protection at home.  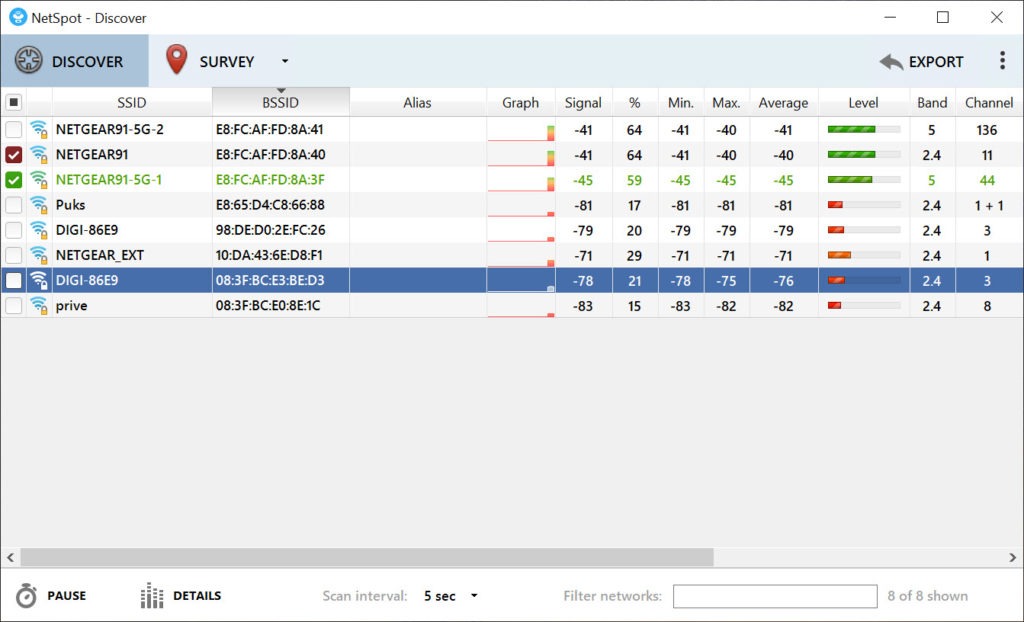
Radiation can be detected in the urine for up to 30 days following LUTATHERA administration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
